Reviewed by Anurag Mishra (Sr. Technical Consultant)

In laboratory practice, accuracy, reliability, and compliance must be ensured. Two core practices that do this are Quality Control (QC) and Quality Assurance (QA). These terms are commonly confused with each other but have different uses. Quality Control is used to identify and correct errors, whereas Quality Assurance is used to avoid errors altogether.
Knowing how they differ ensures that laboratories have uniform performance and comply with regulatory demands. This article will be a detailed guide useful for any industrialist, operator, or businessman. Here we are discussing Quality control vs Quality assurance while comparing characteristics, process, and applications in various industries.
Quality Control (QC) in a laboratory is a process to check that test results are accurate and reliable. QC includes monitoring and checking testing processes for the purpose of spotting and resolving errors, confirming the quality and uniformity of laboratory results. This is important for sustaining confidence in laboratory results and facilitating accurate diagnosis and treatment decisions.
Key characteristics of Quality Control are that it is reactive (identifying defects), product quality-focused, and involves inspection and testing procedures.
Quality control is chiefly a reaction, i.e., it detects flaws once they have been experienced in the manufacturing process. It is aimed at locating and correcting issues instead of stopping them. Instances are product finished checking and functionality testing.
QC operations are product-focused, ensuring that the product complies with pre-set quality requirements. This comprises checking, testing, and analyzing the product to confirm its attributes and performance.
Quality control is dependent on inspection and testing to detect defects and standard deviation. This may be through different means, including visual check, functional testing, and performance testing.
One of the main roles of QC is to ensure that there is consistent quality between production runs. This guarantees that all products are of the same quality level, ensuring reliability and customer satisfaction. In industries such as packaging, maintaining these standards involves periodic testing on a Bursting Strength Tester to verify that materials consistently meet strength requirements across batches.
By catching defects early, QC avoids expensive rework, waste, and even product recall. Early detection reduces the resources' impact and lowers overall production expense.
QC enables companies to comply with industry regulations and stay clear of legal consequences of faulty products. QC ensures conformity to standards and has documentation for audits.
Quality control is the systematic process of verifying that products or services are following predetermined quality standards and specifications. It entails a sequence of steps aimed at detecting, preventing, and correcting defects and variations, ultimately leading to consistency and reliability.
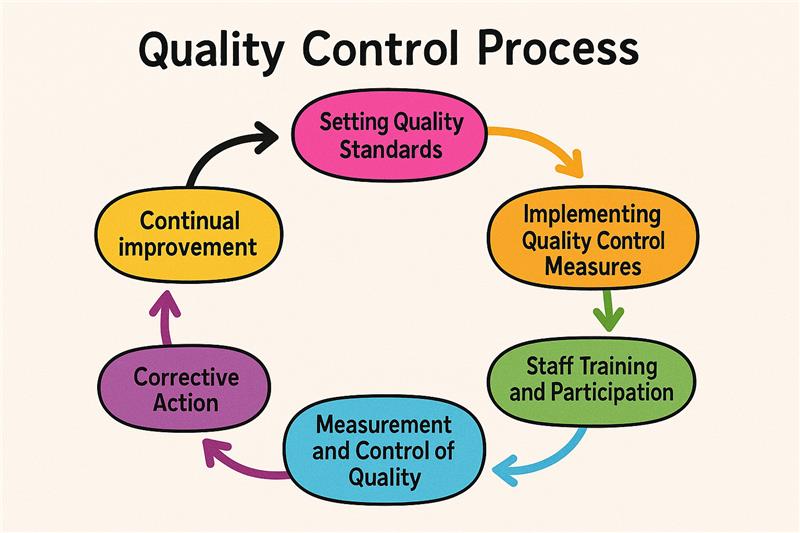
Quality control starts with establishing clear standards and criteria, usually based on customer requirements, industry regulations, or internal objectives. These standards set expectations for raw materials, manufacturing, and end-products.
Once standards are established, organizations implement measures such as inspections and testing during production to follow those standards. This includes the inspection of materials at the beginning, during processing, and before shipping of finished products, with mechanical properties verified through a Universal Testing Machine (UTM) to ensure compliance with quality benchmarks.
The individuals concerned are essential to a successful quality control system. Training staff members in quality principles, tools, and techniques makes them aware of requirements and able to solve problems.
Ongoing monitoring with instruments such as control charts and statistical process control (SPC) is essential in ensuring quality. Data analysis aids in detecting problems and avoiding escalation.
Correcting the causes of quality issues and applying remedies eliminates recurrence. This quick reaction ensures the final product is up to standard and enhances efficiency.
Quality control is a continuous improvement process. This entails acquiring customer input, data analysis, and revising standards to keep up with technology and trends in the industry. Refining the process continually enables companies to remain competitive and produce top-notch results.
Quality Assurance (QA) within the laboratory environment is a well-organized and complete system of procedures, standards, and practices. It aimed at ensuring uniform accuracy, reliability, and validity of all laboratory procedures, right from handling samples to reporting results.
QA includes a series of procedures and processes aimed at reducing errors and ensuring the integrity of laboratory testing. Quality assurance covers all, from setting up standard operating procedures to training staff, calibration of equipment, and continuous monitoring and auditing.
Quality Assurance (QA) is a methodology aimed at avoiding defects and assuring that products or services consistently meet specified requirements, standards, and customer expectations in the entire development or production process.
The following are the main characteristics of Quality Assurance:
QA is dedicated to the development and execution of reliable processes and procedures for maintaining consistent product quality. It places heavy emphasis on documenting and strictly adhering to these processes step by step, from design through delivery.
QA seeks to correct defects and errors prior to their occurrence, instead of identifying and fixing them once they have occurred. This entails conducting quality checks, reviews, and audits along the entire development life cycle.
QA is not a one-time effort but a continuous process of evaluation, improvement, and optimization of procedures and practices. It incorporates taking feedback, data analysis, and determining aspects to improve to increase overall quality in the long term.
QA focuses on knowing and fulfilling customer requirements and expectations. It facilitates the end product or service to match customer needs and provide a smooth and satisfying user experience.
QA relies on data and information to assess quality, identify issues, track performance, and make informed decisions about improvements. It involves using metrics, conducting audits, and gathering feedback to objectively evaluate quality and make data-driven decisions.
QA incorporates quality management practices and principles into an organization's overall management system. It makes sure that quality goals are synchronized with business objectives and backed by the required resources, policies, and procedures.
Quality Assurance (QA) is a systematic and proactive process aimed at anticipating defects in products, services, or processes to ensure they conform to specified quality requirements and customer expectations.
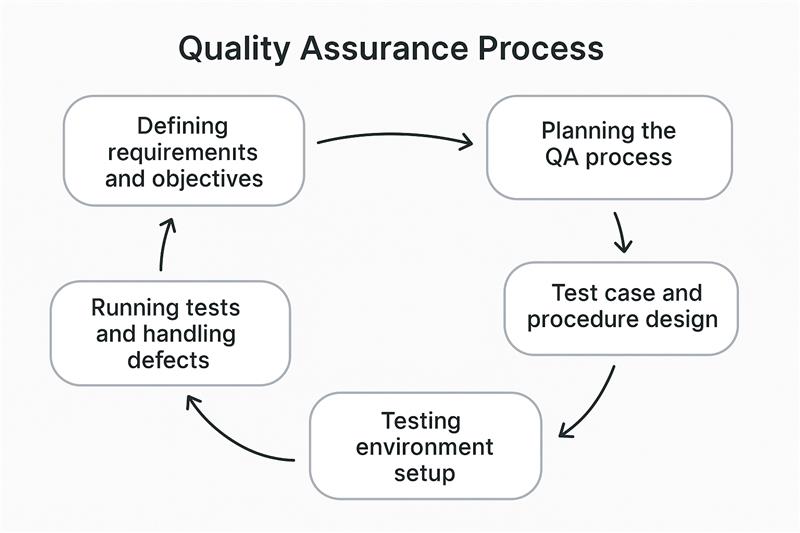
The process starts with the clear identification of the quality requirements and standards for the product, service, or process. These are functional (what it must do) and non-functional (performance, security, usability) requirements.
Create an overall quality management plan that specifies how to achieve the identified quality standards. This involves specifying the test scope, resources (tools, people), setting timelines, and writing the testing strategy.
According to the requirements and test plan, detailed test cases and procedures are designed by the QA team. These outline the precise testing conditions, inputs, steps, and expected results in order to check for functionality, performance, and more. Test scenarios and scripts are planned, and if necessary, automation scripts are designed.
A special testing environment is set up to closely simulate the production environment. This includes hardware configuration, software configuration, network settings, and other items for precise and dependable test outcomes.
The QA team runs the scheduled test cases within the set-up environment, carrying out both manual and automated testing when necessary. Any issues or defects found are recorded and returned to the development team for fixing.
While quality control checks that laboratory results are correct by finding and correcting faults, Quality assurance validates that the process towards those results is not faulty and reliable. QC is reactive and product-focused, whereas QA is proactive and process-focused. Both are necessary to ensure laboratory standards.
| Parameters | Quality Assurance | Quality Control |
| Objective | QA ensures confidence that the desired quality standards will be met. | QC is concerned with achieving the required quality. |
| Technique | QA involves the systematic management of quality. | QC is the process used to inspect and verify quality. |
| Phase Involved? | QA takes place during the development phase. | QC is not involved in the development phase. |
| Tool Type | Managerial tool | Corrective tool |
| Process/product-oriented | QA is process-oriented | QC is product-oriented |
| Goal | Quality assurance aims to eliminate defects before they occur. | Quality Control aims to identify defects and improve them. |
| Execution order | QA is performed before QC | QC is performed after the quality assurance activity is completed. |
| Technique type | Preventive technique | Corrective technique |
| Focus | The main focus is on the intermediate process. | Its main focus is on the final product. |
| Example | Verification | Validation |
Both Quality Control and Quality Assurance play critical roles in laboratory quality management. QC verifies that the results of testing are up to standard by flagging errors, and QA puts in place sound processes to avoid these errors. Both of them constitute an integrated system of ensuring accuracy, reliability, and conformity in the lab operations. For quality control and quality assurance tools for your industry, you can Get in Touch with Testronix Instruments and get high-quality testing equipment at the best price in India and other locations.